[ad_1]
New Delhi: As Covid makes a resurgence in parts of the world driven by a new Omicron sub-variant, India too is updating its pandemic playbook.
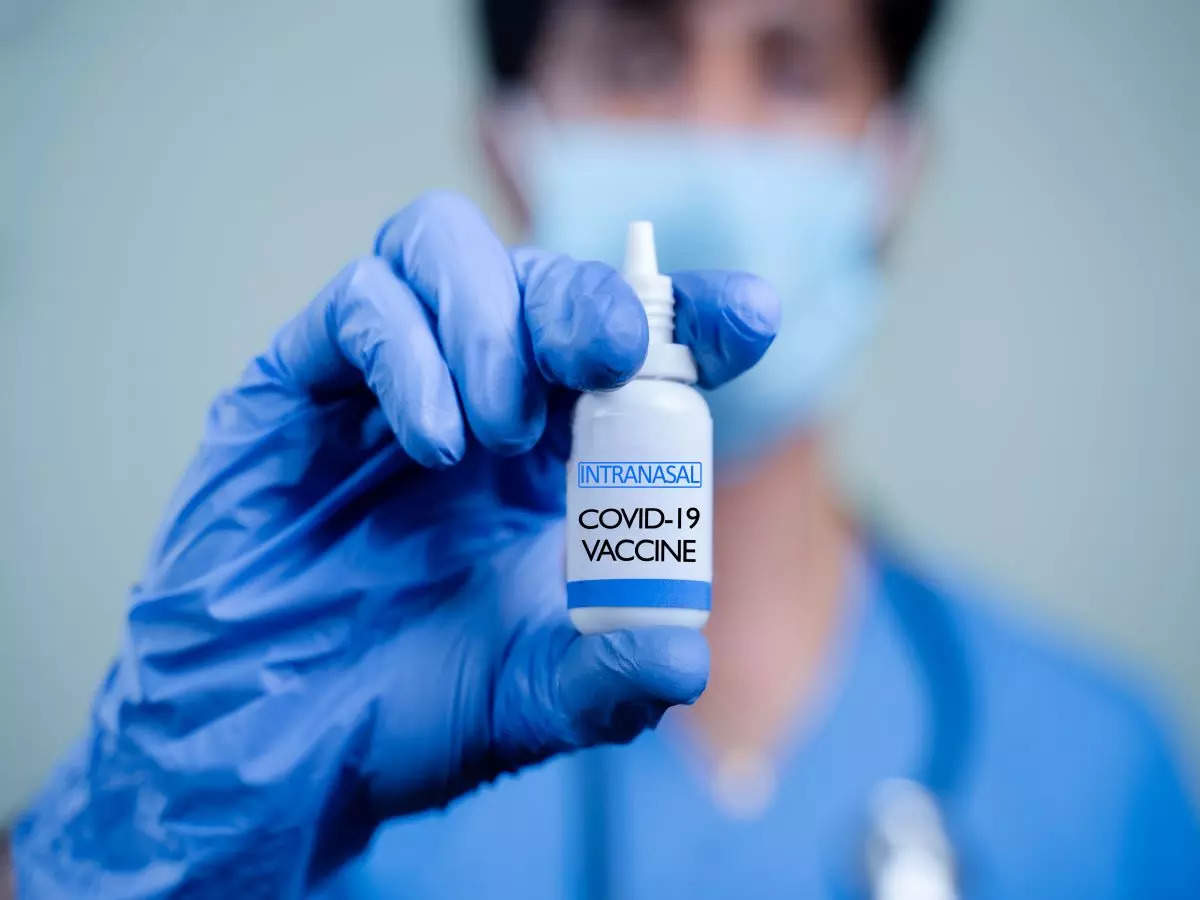
Bharat Biotech‘s intranasal Covid vaccine has been approved by the Union Health Ministry for inclusion in the vaccination programme as a booster dose for those above 18 years of age, official sources said on Thursday.
The needle-free vaccine will be available at private centres. It will be introduced on the Co-WIN platform on Friday evening, they said.
The nasal vaccine BBV154 received approval of the Drugs Controller General of India in November for restricted use in an emergency situation for those above 18 as a heterologous booster dose.
According to Bharat Biotech, the intranasal vaccine has several advantages because the nasal route has excellent potential for vaccination due to the organized immune systems of the nasal mucosa. It is also non-invasive and needle-free and has ease of administration since it does not require trained healthcare workers.
Prime Minister Narendra Modi had on Thursday cautioned people against complacency and urged them to wear masks in crowded places, while also directing officials to strengthen surveillance measures, especially at international airports.
Union Health Minister Mansukh Mandaviya chaired a high-level meeting on Wednesday to review the COVID-19 situation in India and the preparedness of the public health system for surveillance, containment, and management of COVID-19. He urged people to follow COVID-appropriate behaviour and get vaccinated against the virus.
Emphasizing that COVID-19 is not over yet, he asked the officials to be fully geared up and strengthen surveillance.
Mandaviya has already directed the officials to be fully geared up and strengthen the surveillance system for whole genome sequencing of positive case samples to track the variants through the Indian SARS-CoV-2 Genomics Consortium (INSACOG) network to ensure the timely detection of newer variants, if any, circulating in the country.
[ad_2]
Source link
